Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Zr determination with isotope dilution ICP-MS
Zr determination with isotope dilution ICP-MSAsai, Shiho; Hanzawa, Yukiko; Konda, Miki; Suzuki, Daisuke; Magara, Masaaki; Kimura, Takaumi; Ishihara, Ryo*; Saito, Kyoichi*; Yamada, Shinsuke*; Hirota, Hideyuki*
Talanta, 185, p.98 - 105, 2018/08
Times Cited Count:8 Percentile:31.52(Chemistry, Analytical)Estimating the risks associated with radiation from long-lived fission products (LLFP) in radioactive waste is essential to ensure the long-term safety of potential disposal sites. In this study, the amount of  Zr, a LLFP, was determined by ICP-MS after separating Zr from a spent nuclear fuel solution using a microvolume anion-exchange cartridge (TEDA cartridge). The TEDA cartridge achieved highly selective separation of Zr regardless of its small bed volume of 0.08 cm
Zr, a LLFP, was determined by ICP-MS after separating Zr from a spent nuclear fuel solution using a microvolume anion-exchange cartridge (TEDA cartridge). The TEDA cartridge achieved highly selective separation of Zr regardless of its small bed volume of 0.08 cm . The time taken to complete the Zr separation was 1.2 min with a flow rate of 1.5 mL/min, which was 10 times faster than that for a conventional anion-exchange resin column. Almost all the other elements were removed, leading to accurate measurement of
. The time taken to complete the Zr separation was 1.2 min with a flow rate of 1.5 mL/min, which was 10 times faster than that for a conventional anion-exchange resin column. Almost all the other elements were removed, leading to accurate measurement of  Zr. The result connects experimental value to theoretical prediction provided by ORIGEN2, which requires verification. With the measured value, we demonstrated that the theoretical value is reliable enough to estimate radiation risks.
Zr. The result connects experimental value to theoretical prediction provided by ORIGEN2, which requires verification. With the measured value, we demonstrated that the theoretical value is reliable enough to estimate radiation risks.
Miyamoto, Yutaka; Yasuda, Kenichiro
Journal of Nuclear and Radiochemical Sciences (Internet), 18, p.13 - 15, 2018/07
A sequential separation technique using an anion-exchange column developed in the previous works have the potential to completely separate picograms of Am from the lanthanides using mixtures of acetic acid, hydrochloric acid, and nitric acid as the eluents, without any functional ligands or special columns. This experimental result implies that ultra-trace actinides, including Am, Pu, U, and Th in environmental samples can be sequentially separated by combination of these mixed-media eluents and an anion exchange column.
 O/alcohol mixed media by time-resolved laser-induced fluorescence spectroscopy
O/alcohol mixed media by time-resolved laser-induced fluorescence spectroscopyArisaka, Makoto*; Kimura, Takaumi; Suganuma, Hideo*; Yoshida, Zenko
Journal of Radioanalytical and Nuclear Chemistry, 255(2), p.385 - 389, 2003/02
Times Cited Count:5 Percentile:36.77(Chemistry, Analytical)no abstracts in English
Arisaka, Makoto*; Kimura, Takaumi; Suganuma, Hideo*; Yoshida, Zenko
Radiochimica Acta, 90(4), p.193 - 197, 2002/05
Times Cited Count:14 Percentile:65.5(Chemistry, Inorganic & Nuclear)no abstracts in English
Osaka, Masahiko; Koyama, Shinichi; Mitsugashira, Toshiaki; Morozumi, Katsufumi; Namekawa, Takashi
JNC TN9400 2000-058, 49 Pages, 2000/04
The analytical technique for Cm contained in a MOX FUEL was developed and analysis of Cm contained in irradiated fuel of experimental fast reactor "JOYO" was carried out, to contribute to evaluation of transmutation characteristics of MA nuclide in the fast reactor. The procedure of ion-exchange separation of Cm with nitric acid-methanol mixed media essential for the isotopic analysis in irradiated MOX fuel was adopted considering for being rapid and easy. The fundamental test to grasp separation characteristics of this procedure, such as Cm elution position and separation capacity between Cm and Am or Eu, was carried out. ln applying this procedure to the analysis of Cm contained in actual specimen, separation condition was evaluated and optimized, and the procedure consist of impurity removal and Am removal process was devised. This procedure resulted in high recovery rate of Cm and high removal rate of Am and impurity which becomes a problem in sample handling and mass-spectrometry such as Eu and Cs. The Cm separation test from irradiated MOX fuel was carried out using this technique, and Cm isotopic ratio analysis was enabled. The analytical technique for Cm contained in irradiated MOX fuel was established using the procedure of ion-exchange separation with nitric acid-methanol mixed media. The analysis of Cm contained in irradiated MOX fuel of experimental fast reactor "Joyo" was carried out. As a result, it was revealed from measured data that Cm content rate was 1.4 4.0
4.0 lO
lO atom%, small amount of
atom%, small amount of 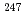 Cm was generated and Cm isotopic ratio was constant above burn-up 60GWd/t.
Cm was generated and Cm isotopic ratio was constant above burn-up 60GWd/t.
Kato, Kaneharu; Ito, Mitsuo; Watanabe, Kazuo
Fresenius Journal of Analytical Chemistry, 366(1), p.55 - 58, 2000/01
no abstracts in English
Sumiya, Shuichi; Hayashi, Naomi; ; Narita, Osamu
PNC TN8430 91-001, 45 Pages, 1990/12
A radioanalytical method for low level samarium-151(Sm-151) and promethium-147(Pm-147) in environmental samples has been studied for the environmental assessment around nuclear facilities. In this study, we use the separation method with high performance liquid chromatography (HPLC) to determine Sm-151 and Pm-147 in environmental samples such as sea sediments and marine organisms. Samarium-151 and Pm-147 in environmental samples are coprecipitated with other lanthanoids after adding neodymium(Nd). These nuclides are purified by anion exchange methods in methanol-mineral acid media. After the purification, Sm-151 and Pm-147 are separated with HPLC in lactic acid-sodium hydroxide media, and determined with liquid scintillation counting, respectively. The Nd is determined by inductively coupled plasma atomic emission spectrometry (ICP-AES) to correct chemical recoveries of these nuclides. The detection limits for Sm-151 and Pm-147 in this method are about 0.01Bq/sample.
; Kubota, Masumitsu
Journal of Nuclear Science and Technology, 26(11), p.1038 - 1044, 1989/11
Times Cited Count:24 Percentile:88.99(Nuclear Science & Technology)no abstracts in English
 BO
BO media
media; ; ; *
Bulletin of the Chemical Society of Japan, 57(6), p.1487 - 1490, 1984/00
Times Cited Count:2 Percentile:24.29(Chemistry, Multidisciplinary)no abstracts in English
 BO
BO media
mediaBulletin of the Chemical Society of Japan, 55, p.1824 - 1830, 1982/00
Times Cited Count:8 Percentile:54.23(Chemistry, Multidisciplinary)no abstracts in English
;
Bunseki Kagaku, 24(5), p.303 - 307, 1975/05
no abstracts in English
;
Journal of Inorganic and Nuclear Chemistry, 35(7), p.2592 - 2594, 1973/07
Times Cited Count:5no abstracts in English
Nihon Genshiryoku Gakkai-Shi, 4(3), p.154 - 160, 1962/00
no abstracts in English
; ; *
Bunseki Kagaku, 7, P. 720, 1958/00
no abstracts in English
; ; *
Bunseki Kagaku, 7, P. 721, 1958/00
no abstracts in English
Sato, Makoto; Ichimura, Seiji; Suyama, Kenya; Tonoike, Kotaro; Kamohara, Keiko*; Suzuki, Norihisa*
no journal, ,
no abstracts in English
Asai, Shiho; Hanzawa, Yukiko; Konda, Miki; Suzuki, Daisuke; Magara, Masaaki; Kimura, Takaumi; Ishihara, Ryo*; Saito, Kyoichi*; Yamada, Shinsuke*; Hirota, Hideyuki*
no journal, ,
 Zr is a long-lived fission product which can be found in spent nuclear fuel and HLW. The estimation of the
Zr is a long-lived fission product which can be found in spent nuclear fuel and HLW. The estimation of the Zr content is indispensable to achieve a safety disposal of HLW because
Zr content is indispensable to achieve a safety disposal of HLW because  Zr is predicted to be one of the major contributors to radiation dose. However, only a few measured
Zr is predicted to be one of the major contributors to radiation dose. However, only a few measured  Zr values have been reported, leading to a high demand for development of an efficient analytical method. Our group has been prepared a new porous filter cartridge which has densely bound ion-exchanger onto the pore surface of the filter, enabling a high-capacity and rapid adsorption. In order to apply this filter cartridge to a pretreatment for the
Zr values have been reported, leading to a high demand for development of an efficient analytical method. Our group has been prepared a new porous filter cartridge which has densely bound ion-exchanger onto the pore surface of the filter, enabling a high-capacity and rapid adsorption. In order to apply this filter cartridge to a pretreatment for the  Zr measurement with ICP-MS, an elution profiles of Zr and the other coexisting elements were examined. According to the resultant separation conditions, Zr in a spent nuclear fuel sample was successfully separated. The measured
Zr measurement with ICP-MS, an elution profiles of Zr and the other coexisting elements were examined. According to the resultant separation conditions, Zr in a spent nuclear fuel sample was successfully separated. The measured  Zr content is 98.2
Zr content is 98.2  5.1 ng, which agrees with the theoretical value.
5.1 ng, which agrees with the theoretical value.
Asai, Shiho; Hanzawa, Yukiko; Konda, Miki; Suzuki, Daisuke; Magara, Masaaki; Kimura, Takaumi
no journal, ,
Various Zr isotopes generated by U fission are found in spent fuel and HLW. Among them,  Zr which has a long half-life of 1.5
Zr which has a long half-life of 1.5 10
10 y has a potential to contribute to radiation dose over an extended period of time after the implementation of HLW disposal. The inventory estimation of
y has a potential to contribute to radiation dose over an extended period of time after the implementation of HLW disposal. The inventory estimation of  Zr in HLW confirmed by measured data is the key to realize a safe and cost-efficient disposal. In this study, a simple and robust analytical technique for the determination of
Zr in HLW confirmed by measured data is the key to realize a safe and cost-efficient disposal. In this study, a simple and robust analytical technique for the determination of  Zr based on ion-exchange chromatography combined with ICP-MS was developed. Interference-free measurement was achieved by a single anion-exchange step, removing Sr, Nb, and Mo which would cause spectral interferences. Additionally, major component U and radioactive components, such as Cs, Ba, and Pu, were also removed concurrently. Concentration of
Zr based on ion-exchange chromatography combined with ICP-MS was developed. Interference-free measurement was achieved by a single anion-exchange step, removing Sr, Nb, and Mo which would cause spectral interferences. Additionally, major component U and radioactive components, such as Cs, Ba, and Pu, were also removed concurrently. Concentration of  Zr was readily calculated with measured isotope ratios of
Zr was readily calculated with measured isotope ratios of  Zr/
Zr/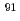 Zr in the sample and natural Zr-spiked sample with sufficient accuracy.
Zr in the sample and natural Zr-spiked sample with sufficient accuracy.
Aono, Ryuji; Sato, Yoshiyuki; Ishimori, Kenichiro; Kameo, Yutaka
no journal, ,
no abstracts in English